Beijing-Sana
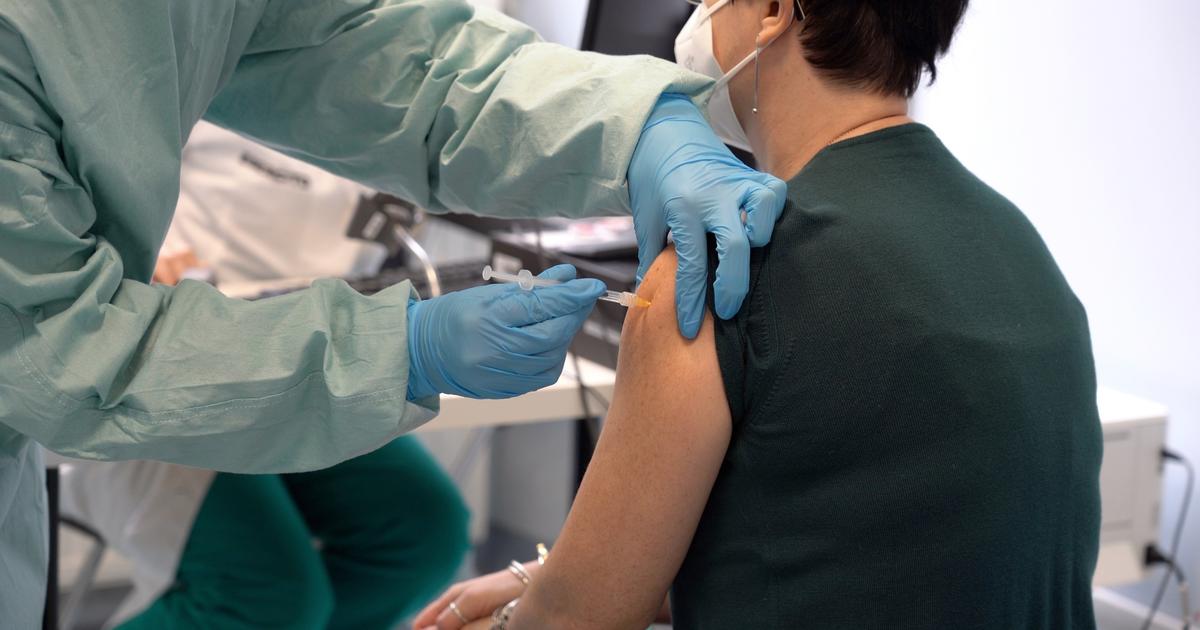
Four Chinese vaccines against "Covid 19" have begun the third clinical trial phase, according to the Joint Prevention and Control Mechanism of the State Council of China.
And Xinhua News Agency reported that some third phase experiments are expected to be completed in early September, while preliminary data is expected to appear in November.
The third phase of clinical trials usually involves the participation of thousands of people to verify the safety and efficacy of vaccines, which are the two main guarantees for approval for placing on the market.
And it has been approved to conduct the third clinical trial phase on two inactivated anti-"Covid 19" vaccines developed by the Chinese National Biotech Group in a number of countries in the Middle East and South America with the participation of more than 30 thousand people. In addition, an inactivated anti-disease vaccine developed by the "Sinovac Biotech" Company Ltd. is subject to the stage. The third of the experiments in a number of countries in South America and Southeast Asia, while another vaccine made of the modified adenovirus as a vector is subject to the third phase of the trials as well.
According to guidelines for the clinical evaluation of "Covid 19" vaccines recently issued by the Chinese Medical Products Administration, the vaccine must provide immunity for at least six months and preferably more than a year.