Social News
Written by: Huang Weilun
2020-09-04 20:34
Last update date: 2020-09-04 20:34
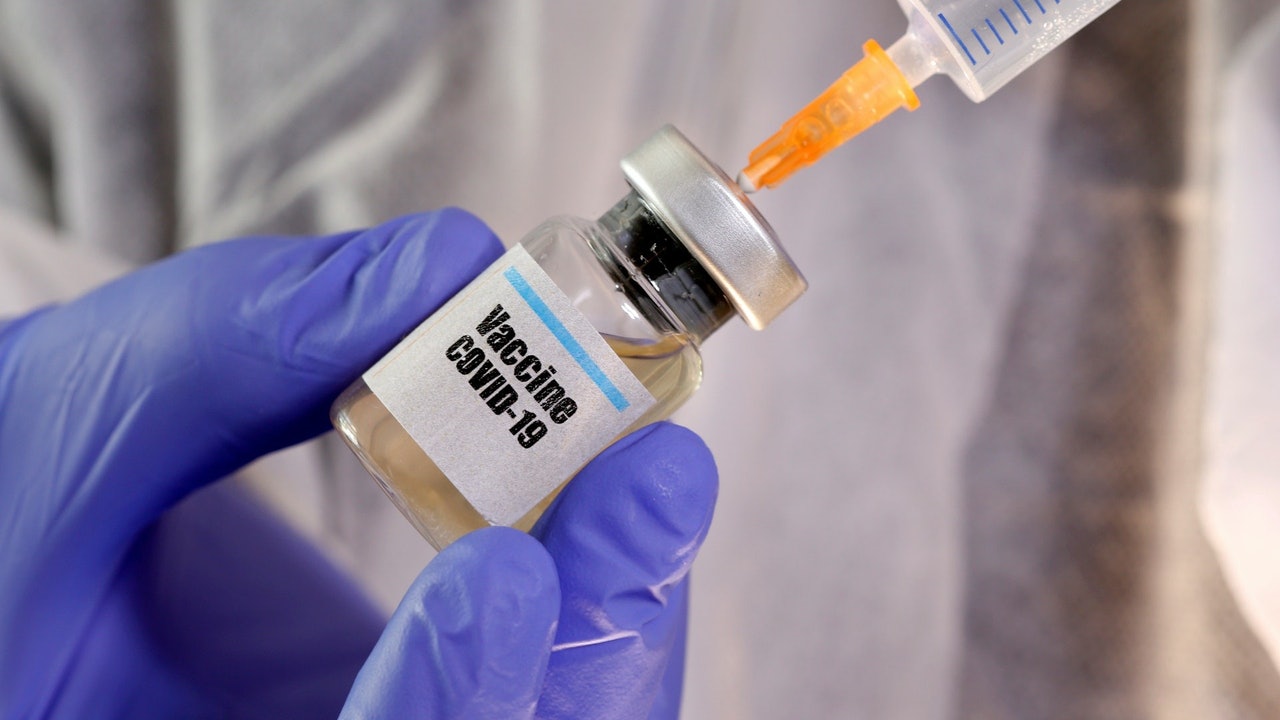
New crown pneumonia rages the world, and many countries have developed vaccines.
The government announced today (4th) that it has submitted a letter of intent to the Global Access Mechanism for COVID-19 Vaccine (COVAX Mechanism) as part of Hong Kong's vaccine procurement strategy.
A spokesman for the Food and Health Bureau said that participation in the COVAX mechanism can provide Hong Kong with a certain degree of safety net.
In addition, the government stated that it will directly purchase more vaccines from vaccine manufacturers in various regions through pre-purchase agreements to seek additional vaccine supplies.
▼From September 4th, some premises are still closed to relax the measures▼
+18
+18
+18
The COVAX mechanism is led by the Global Alliance for Vaccines and Immunization (Gavi), Coalition for Epidemic Preparedness Innovations (CEPI) and the World Health Organization. It aims to promote the development and production of new coronavirus vaccines and ensure fairness around the world To distribute vaccines and protect the most vulnerable groups, the goal is to deliver vaccines to participating countries or economies by the end of 2021.
The government stated that although the COVAX mechanism can only ensure that the amount of vaccines can cover some Hong Kong citizens, the government will, based on scientific evidence and clinical data, consult the relevant scientific committees under the Department of Health, and use pre-purchase agreements to direct vaccine manufacturers around the country. Purchase more vaccines to seek additional supplies.
The Department of Health is seeking information on vaccine research and development or preliminary production from various places, including the Mainland, for research and discussion with the Scientific Committee.
▼Yuan Guoyong demonstrates and compares the advantages and disadvantages of the four detection sampling methods▼
+2
+2
+2
A spokesperson for the Food and Health Bureau said that although according to the WHO, no vaccine has been approved for human use by regulatory agencies, it is expected that global competition will be fierce when effective vaccines are available. Therefore, Hong Kong must act as soon as possible to serve the public. Reserve potential vaccines in time, including participation in the COVAX mechanism, which can provide Hong Kong with a certain degree of safety net.
A government spokesperson also stated that it will continue to adopt a "two-pronged" strategy to purchase vaccines that meet the safety, efficacy and quality requirements for the public to protect their health.
[New Coronary Pneumonia] The US vaccine was approved in October?
Fauci: unlikely but not impossible
[New Coronary Pneumonia] The United States: Will not join the WHO-related vaccine development
[New Coronary Pneumonia] Kang Sino: Expected to see preliminary results of vaccine phase III clinical trials in 3 to 6 months
[New Coronary Pneumonia] WHO calls on all countries to maintain epidemic prevention measures not to approve the use of vaccines easily
What do you know|Results of vaccine clinical trials in various countries: Which one is more effective?
What are the side effects?
The vaccine game is becoming increasingly fierce, Chinese companies and many countries discuss the use of military research and development vaccines
New crown vaccine vaccine new crown pneumonia new crown pneumonia