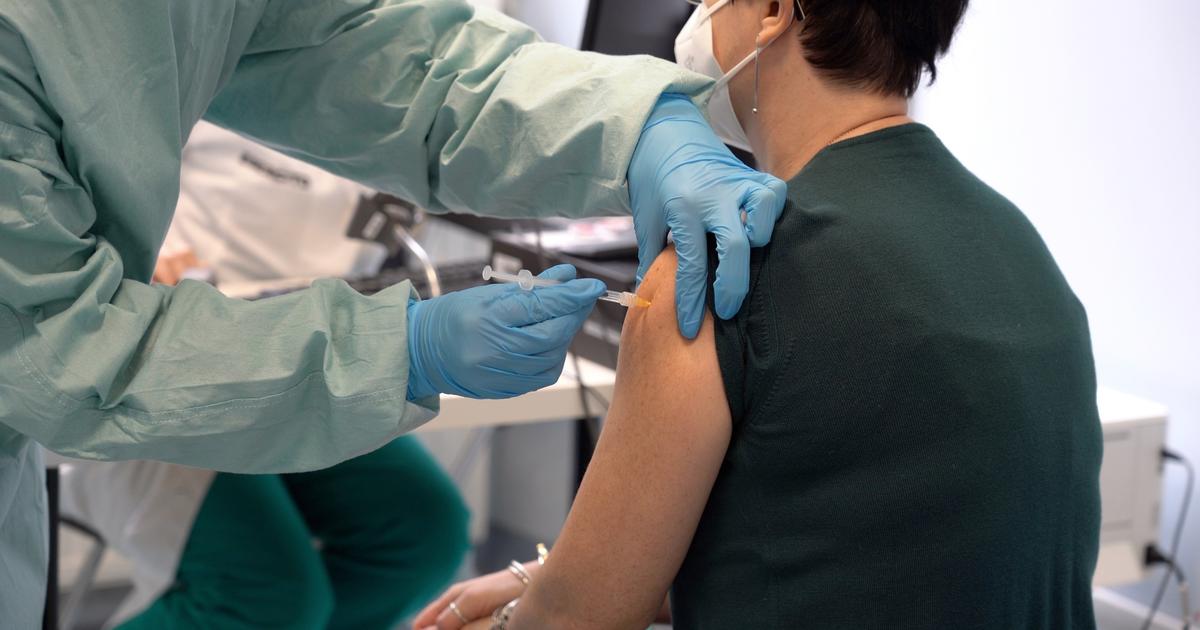
Less than ten weeks after China published the genetic code for the SARS-Cov-2 coronavirus responsible for the current pandemic, a Cambridge, Massachusetts-based biotech Moderna launched the first phase of its candidate's clinical trial. vaccine. A record! If the young company and other manufacturers in the process of launching their own trials have been able to move so quickly, it is because of a different approach based on the genetics of the virus.
Read also: Coronavirus: "Our vaccine could be injected to caregivers in the fall"
But manufacturers know this, despite the health emergency, many obstacles still stand before them and it will take several months before a vaccine can be marketed. However, the urgency is there: everyone wants to be ready for the possible return of the coronavirus after the current epidemic wave. Manufacturers must also move quickly to conduct clinical trials with vaccine candidates while the virus is circulating, because if the pandemic declines quickly, which would be good for threatened populations, it
This article is for subscribers only. You have 70% left to discover.
Subscribe: € 1 the first month
cancellable at any time
Enter your emailAlready subscribed? Log in