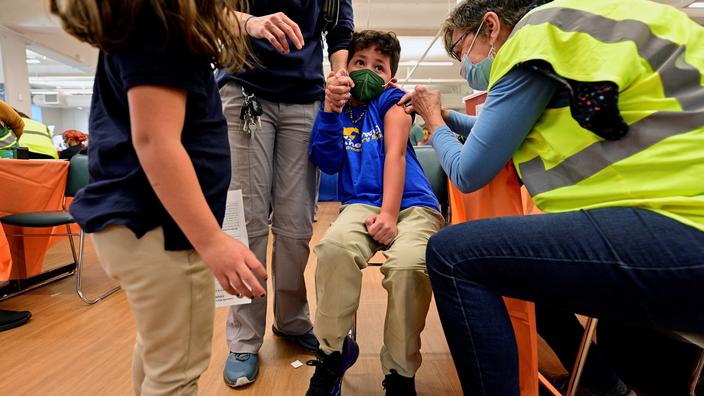
Canadian health authorities announced on Friday that they are allowing the use of the Pfizer / BioNTech anti-Covid vaccine for children aged 5 to 11.
"
After a complete independent scientific review, the Ministry (of Health, editor's note) determined that the benefits of this vaccine for children aged 5 to 11 outweigh the risks,
" the authorities said in a statement.
Lower dosage
The vaccine dosage for 5 to 11 year olds was lowered to 10 micrograms per injection, three times less than the standard dose.
The injections will have to be done "
three weeks apart,
" said Health Canada, which does not disclose for the moment the start date of the vaccination campaign.
In the clinical trial, the immune response of children aged 5 to 11 was comparable to that of people aged 16 to 25.
The vaccine was 90.7% effective in preventing Covid-19 in children aged 5 to 11, and no serious side effects were identified, the ministry added.
The Pfizer / BioNTech alliance had filed its application for authorization on November 18, based on data from trials conducted on 2,268 children of the age group.
Read alsoCovid-19: a 3rd dose of the Pfizer / BioNTech vaccine effective at 95.6%
Elsewhere in the world, a small number of countries including Israel and the United States have so far authorized Pfizer / BioNTech's vaccine for children.
China, the United Arab Emirates, Cambodia and Colombia have also started to vaccinate children under 12 but with Chinese vaccines.