health
news
Why should children be vaccinated if the fourth wave of the corona is behind us?
73 experts voted in favor of corona vaccines for children from the age of 5, however 2 voted against, in the simple argument that there is no reason to rush.
We looked at what was behind their claim, and what we had already learned from the rapid vaccination campaign in the US
Tags
Corona
Vaccine for corona
Vaccines
Children
covid-19
Pfizer
Walla!
health
Thursday, 11 November 2021, 11:40 Updated: 12:54
Share on Facebook
Share on WhatsApp
Share on general
Share on general
Share on Twitter
Share on Email
0 comments
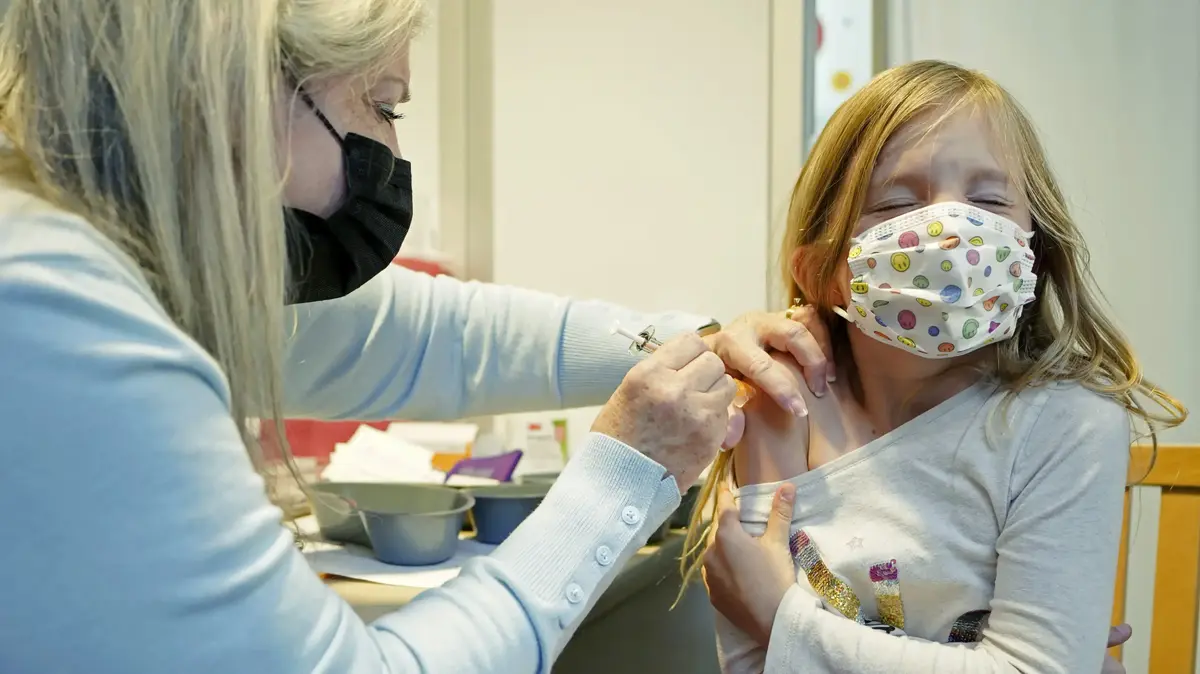
In a sweeping majority - senior Ministry of Health officials approve of vaccinating children between the ages of 5-11 (edited by Aviad Ballali)
Health Ministry experts yesterday (Wednesday) approved the Corona vaccine for children aged 11-5.
The epidemiological team decided to vaccinate children against the virus by a majority of 73 to 2. The opposition of the two experts who voted against was not sweeping, but opposition to the vaccine at this stage.
Their argument is that in the current disease state we have the privilege of waiting for information to be gathered from the world and making the decision to vaccinate later.
More on Walla!
Research has revealed: This is what the corona vaccine really does to the heart muscle
To the full article
The rationale behind this approach is that the fourth wave of the corona plague is now on the wane in Israel, so there is no reason to hurry and vaccinate children, and one can take advantage of the declining morbidity to gather more information from other countries that started vaccinating children. However, health professionals believe that it is precisely this decrease in morbidity that is our chance to succeed in gaining an advantage over the virus that comes back and strikes again and again.
"The experts who voted against may think that there is no point in vaccinating at the moment because there is no risk, that is, there is little morbidity in the country today. Dr. Ella Skalen, Department of Immunology and Microbiology at the Faculty of Medicine of Tel Aviv University.
Wait for more information from other countries that store children?
11-year-old Leo Hahn receives first-degree corona vaccine in Washington, DC (Photo: AP, Ted S. Warren)
Dr. Skellen added that "another reason that may make them want to wait is to see if there are any additional side effects that will be discovered in the children's vaccines in the US, however since in the US we have already vaccinated close to a million children aged 5-11 I do not think there is any point in waiting ".
indeed, at the United States, showing no concern might be justified.
The data show that close to one million children aged 5-11 have already been vaccinated with one dose of the Corona vaccine in the eight days since the vaccine was approved by the FDA and CDC (Center for Disease Control and Prevention) and given the green light to vaccinate younger children.
More on Walla!
The state is no longer willing to pay for medical care for unvaccinated corona patients
In this country, two-year-old toddlers have already been vaccinated against corona
Does B-Cure Laser Really Help With Knee Pain?
It's the truth
By the end of Wednesday, more than 900,000 children in the 5-11 age group had been vaccinated in the US in the first dose of the covid-19 vaccine - the White House revealed this figure last night. Although this is a high and impressive number, it is 3 percent However, a large number of more children are expected to be vaccinated in the coming days and weeks, according to data released by the White House, which includes another 700,000 vaccinations for children that have already been scheduled for the next few days in pharmacies across the United States.
Also, many more children are expected to get the vaccine at their pediatricians ’clinics, hospitals and more.
More on Walla!
Vaccinate or wait?
Experts answer your questions about corona vaccines for children
To the full article
The only vaccine currently approved for young children by the FDA is the Pfizer BioNTech vaccine, which contains the same composition of the adult and adolescent vaccine, except that five to eleven year olds will receive it in a reduced dose - two doses of 10 micrograms, given 3 weeks apart. (Unlike the adult dose, which stands at two doses of 30 mcg).
approval of the emergency provision of Pfizer's vaccine to children under 12 accepted by the FDA in late October, after the company presented data on efficacy and safety are good and most promising clinical trials , Which proved that the vaccine produced a strong and good immune response in young children with very few side effects.
Share on Facebook
Share on WhatsApp
Share on general
Share on general
Share on Twitter
Share on Email
0 comments