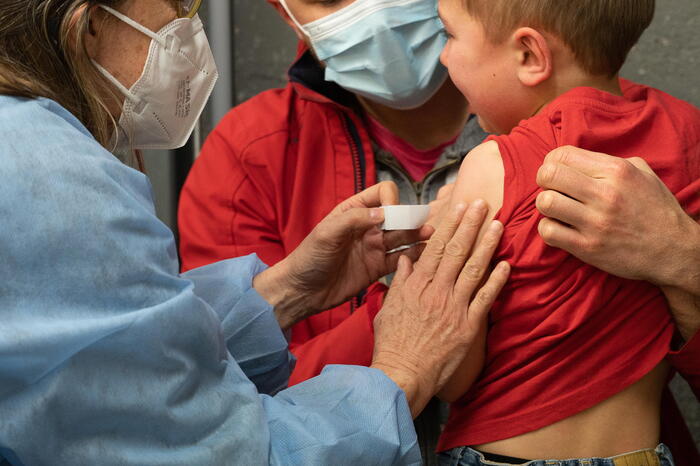
(ANSA) - NEW YORK, MAY 17 - The Food and Drug Administration has authorized Pfizer's Covid vaccine booster for children between 5 and 11 years old.
The administration of the booster can take place at least 5 months after inoculation of the first complete series of the Pfizer vaccine on children.
In the US - out of 28 million children of that age - only 8 million have received the necessary first two doses.
The US Centers for Disease Control and Prevention will most likely confirm the FDA indication.
The meeting of CDC experts to discuss the issue is scheduled for Thursday.
(HANDLE).