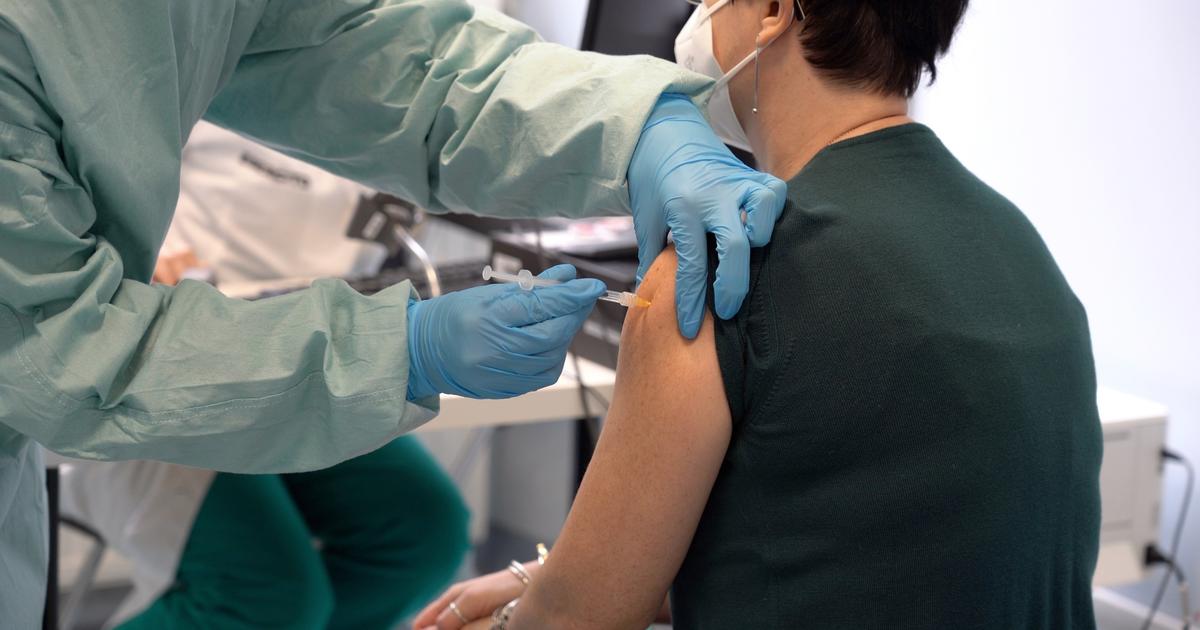
(ANSA) - BRUSSELS, AUGUST 09 - "To best guarantee our common preparation" against Covid in autumn and winter, "Member States must have the necessary tools. This includes vaccines adapted to variants, as and when they will be authorized by the European Agency for medicines "(Ema).
This was stated by the EU Commissioner for Health, Stella Kyriakides, underlining that the agreement between the EU and modern day to re-schedule the deliveries of vaccines against Covid-19 in September "will ensure that countries have access to the drugs they need at the right time. to protect citizens ".
The agreement also covers vaccines adapted to the new BA.4 and BA.5 Omicron variants, pending authorization by the EMA.
"
"The agreement - explains the European Commission - guarantees that, if one or more adapted vaccines receive marketing authorization, the Member States can choose to receive the adapted vaccines under the current contract" and also provides for the delivery of " an additional 15 million doses of "adapted vaccines" subject to marketing authorization within a time frame that would allow the use of these doses for their vaccination campaigns ".
(HANDLE).