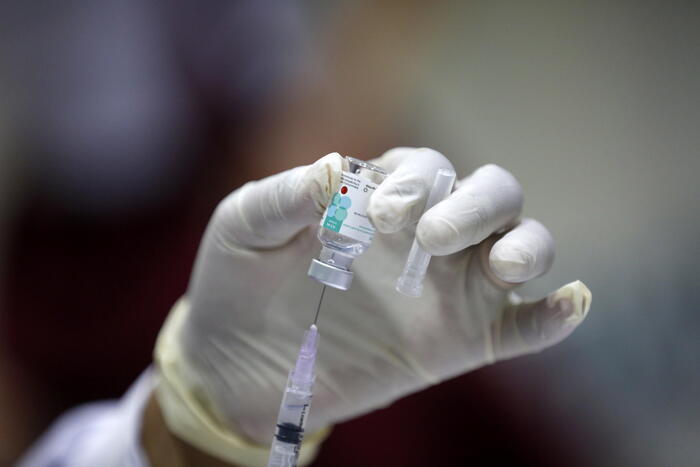
There has been one death in a corona vaccine study in Brazil.
A subject in the clinical study from the pharmaceutical company AstraZeneca has died.
Brasília - A participant in a series of tests conducted by Oxford University in the UK for a vaccine against the coronavirus * has died.
In the tests in Brazil, however, the man should
not have been given
the potential serum, but only a
placebo
, as Brazilian media reported on Wednesday.
The university said an independent expert review concluded there were
no safety issues
and testing could continue.
The case is currently being examined and data on volunteers must be treated confidentially.
Death in corona vaccine study by AstraZeneca
According to media reports, the deceased are a 28-year-old doctor who was active in the fight against the pandemic and who voluntarily participated in the tests for the vaccine.
The man was
infected
with the novel
corona virus
and died as a result of this infection.
Initially, neither the laboratory nor the test centers or the Brazilian health surveillance authority, Anvisa, officially commented on this.
In test series for new drugs or vaccines, it is common to give some of the participants not the agent to be tested, but only a placebo, to compare the effects.
A placebo is a bogus drug that does not contain any drug.
Corona vaccine in phase III
Oxford University is researching
an anti-corona vaccine
together with the British pharmaceutical company
AstraZeneca
.
The tests at Oxford University and AstraZeneca are in the
third
and final phase, which is very complex.
Thousands of people in several countries take part, in Brazil there are around 8,000 so far.
The drug, tested by Oxford University and AstraZeneca, is considered one of the most promising vaccine candidates worldwide.
Vaccines against the coronavirus are being feverishly researched around the globe.
According to the WHO (Draft landscape of COVID-19 candidate vaccines, as of October 19, 2020), around 198 vaccines are being researched around the world - almost ten vaccine candidates are in the third and final test phase.
In Germany, three pharmaceutical companies are working on a corona vaccine: CureVac, BioNTech and IDT Biologika *.
AstraZeneca temporarily halted a clinical trial for its promising corona vaccine in September.
The
vaccine AZD1222
counted until then to the promising candidates among potential corona vaccines.
Should the death of the volunteer be linked to the vaccine, it would be a major blow to the project.
The vaccine is currently being examined by the European Medicines Agency (EMA) in a so-called rolling review process.
In this process, data from the clinical trial is continuously submitted and evaluated.
The EU has already signed a framework agreement with AstraZeneca for the delivery of vaccination doses.
According to the Federal Ministry of Health, Germany accounts for 54 million vaccine doses.
According to the head of department Jens Spahn, that would initially be enough for around 27 million people in this country.
(AFP, dpa) * Merkur.de is part of the Ippen digital network