Peru launched its Covid-19 vaccination campaign on Tuesday with the vaccine developed by the Chinese laboratory Sinopharm, while Argentina gave the green light to the use of the Covishield vaccine, resulting from a transfer of AstraZeneca technology.
Read also: Second wave of Covid-19 in Peru, says government
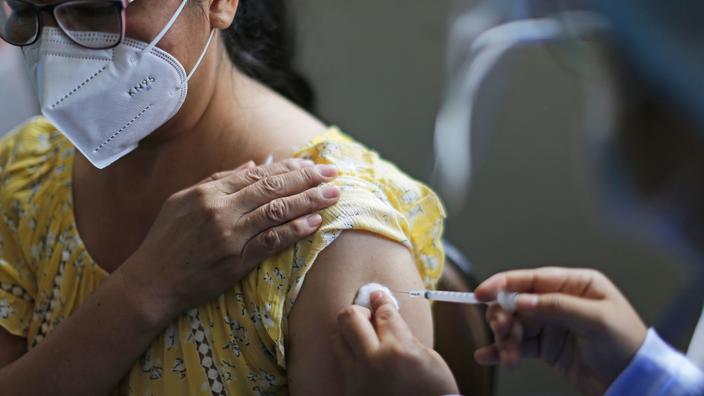
The Peruvian vaccination campaign began Tuesday morning in hospitals in Lima, where several hundred doctors, nurses and health workers received the first dose of the vaccine.
"
We have established a five-day schedule
" to vaccinate all staff, explained the director of San Bartolomé Hospital, Carlos Santillán, after receiving the first dose.
Health workers have paid a heavy price for the pandemic in Peru: 298 doctors and 25 nurses have died since March, according to professional organizations.
By Sunday, the country is expected to have one million doses of Sinopharm's vaccine.
Army planes took off from the Peruvian capital with vaccines intended for health workers in the regions most affected by the second wave of the pandemic, including Huanuco, 350 km northeast of Lima.
Peru, a country of 33 million people, bought 38 million doses from Sinopharm and 20 million of the German-American vaccine Pfizer-BioNTech.
He is also to receive 14 million doses of the British Oxford-AstraZeneca vaccine and 13 million doses through the Covax device of the World Health Organization (WHO).
With more than 1.2 million reported cases, including more than 42,000 fatalities, Peru is one of the countries most bereaved in the world by the pandemic in relation to its population.
For its part, Argentina, which started its vaccination campaign on December 29 with the Russian vaccine Sputnik V, on Tuesday authorized the use of the Covishield vaccine manufactured by the Indian laboratory Serum Institute of India (SII).
This is the vaccine developed by the University of Oxford-AstraZeneca, already approved in Argentina on December 30, manufactured in India from a transfer of technology.
The Serum Institute of India (SII) is producing millions of doses of this vaccine at high speed for India and much of the developing world.
This was to ensure "
that such a transfer (of technology) does not impact the quality, safety and efficacy
" of the product which has already been approved by twelve countries, in addition to the Indian health authority, the Argentine drug authority said.