By Alexander Smith - NBC News
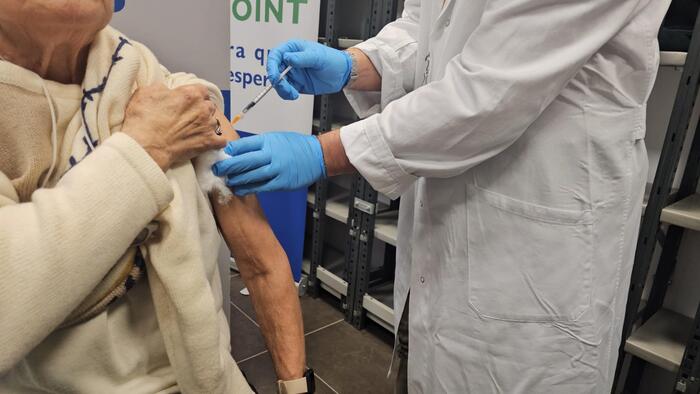
Federal health agencies on Tuesday recommended stopping use of the Johnson & Johnson COVID-19 vaccine after a small number of people experienced "a rare and severe type of blood clot" after receiving the injection.
The Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC) issued a statement recommending "a pause in the use of this vaccine as a precaution ".
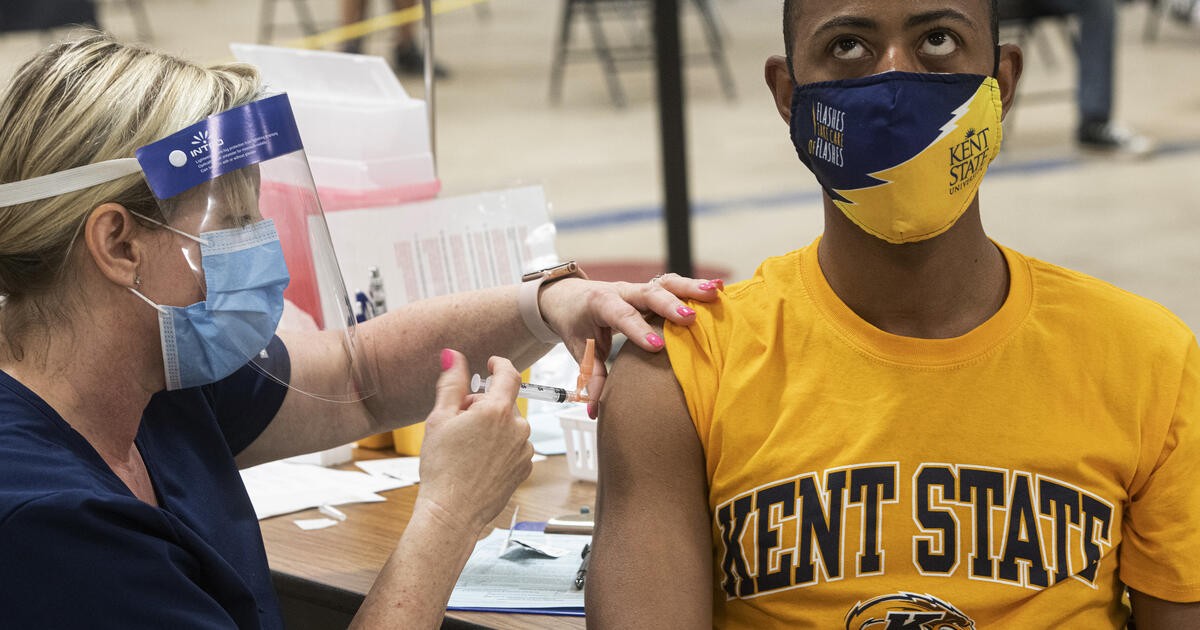
Kent State University student Marz Anderson receives his COVID-19 vaccine from Johnson & Johnson in Kent, Ohio, Thursday, April 8, 2021.AP Photo / Phil Long
About 6.8 million people have received the one-shot vaccine from this company in the United States.
Six have experienced the clot, according to the agencies: "At this time, these adverse events appear to be extremely rare," they added.
News in development.