Are vaccines enough to protect against variants?
0:53
(CNN) -
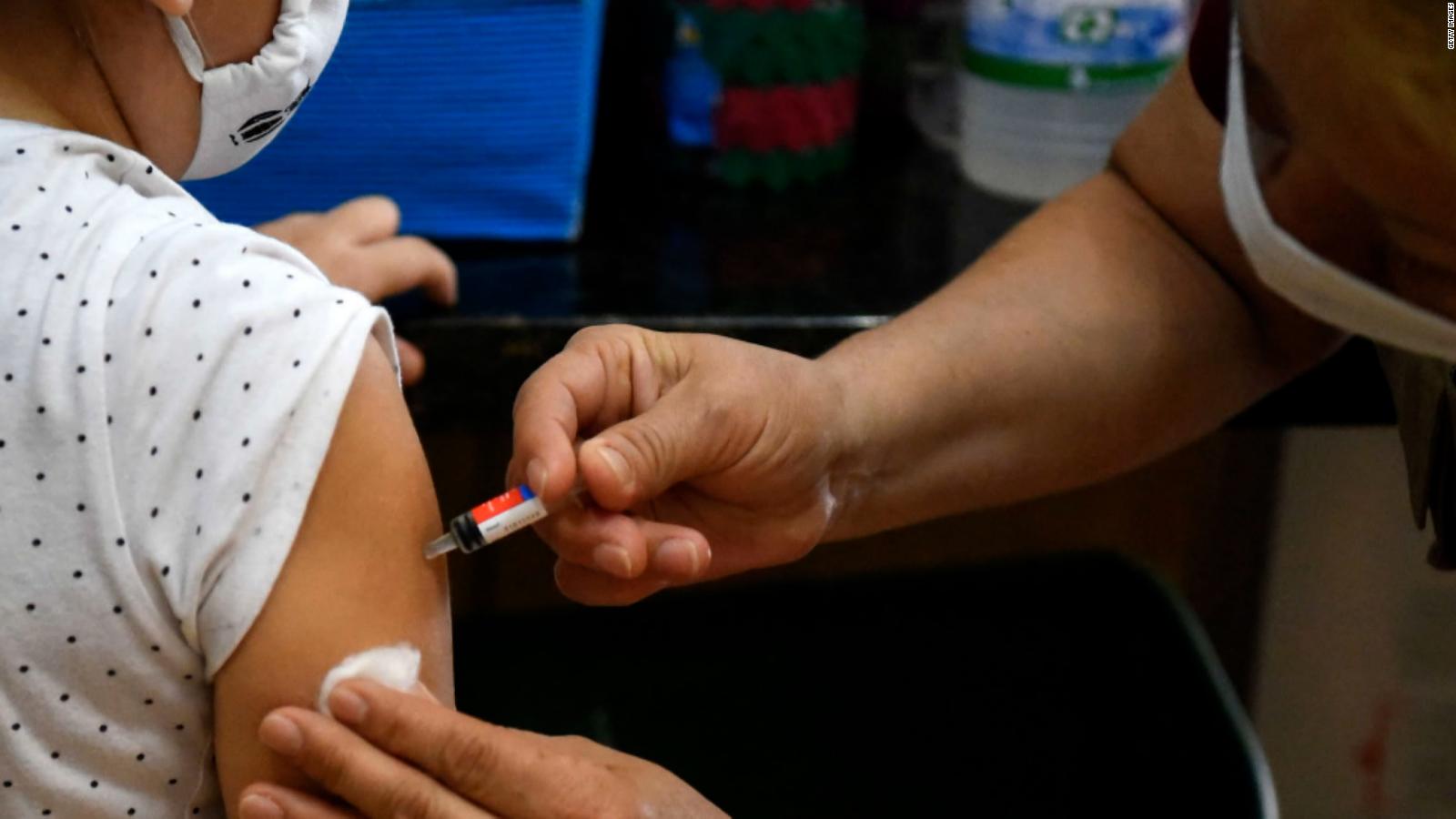
Among the millions of teens who have received the Pfizer / BioNTech coronavirus vaccine, new data shows that the majority of those who have reported adverse effects experienced mild problems, with heart condition myocarditis ranked in the 4 , 3% of all reports.
Data released Friday by the U.S. Centers for Disease Control and Prevention (CDC) reveals that among the 8.9 million adolescents vaccinated between December and July, reports were received. adverse effects of about 1 in 1,000 vaccines.
In total, 8,383 or 90.7% of the reports were for non-serious events and 9.3% for serious events, including death.
No reports of death were determined to be the result of myocarditis.
How does the new CDC guide on masks change the way you take care of yourself against COVID-19?
An expert opinion
According to the report, among the serious notifications the most frequent were chest pain, with 56.4%;
the increase in troponin, which can indicate a heart problem, with 41.7%;
and myocarditis, or inflammation of the heart muscle, with 40.3%.
Starting in June, cases of myocarditis appeared among young people after receiving the vaccine, mainly among men.
That same month, the U.S. Food and Drug Administration added a warning to Moderna and Pfizer / BioNTech's coronavirus vaccine fact sheets.
Adverse effect reports were received from approximately 1 in 1,000 vaccines.
The most common effects
CDC researchers wrote in the new data that, as of July 16, the federal government's Vaccine Adverse Event Reporting System, or VAERS, received 9,246 reports among youth ages 12 and up. 17 years after receiving the Pfizer / BioNTech vaccine, out of about 8.9 million adolescents vaccinated.
The VAERS surveillance system relies on individuals to submit reports and may not be generalizable to the entire vaccinated population.
The most common conditions reported were dizziness, temporary loss of consciousness, and headache.
advertising
Covid-19 vaccines are not enough to stop variants, study finds
"The findings summarized in this report are consistent with the safety data observed in the Pfizer-BioNTech preauthorization trials after vaccination among people 12 to 25 years of age, with the exception of myocarditis, a serious adverse event detected in the control. post-authorization security, "the CDC researchers wrote in the report.
"Local and systemic reactions after vaccination with the Pfizer-BioNTech vaccine were frequently reported by adolescents aged 12 to 17 years to US vaccine safety surveillance systems, especially after dose 2 "the researchers wrote.
"A small proportion of these reactions are consistent with myocarditis."
CDCVaccines against covid-19