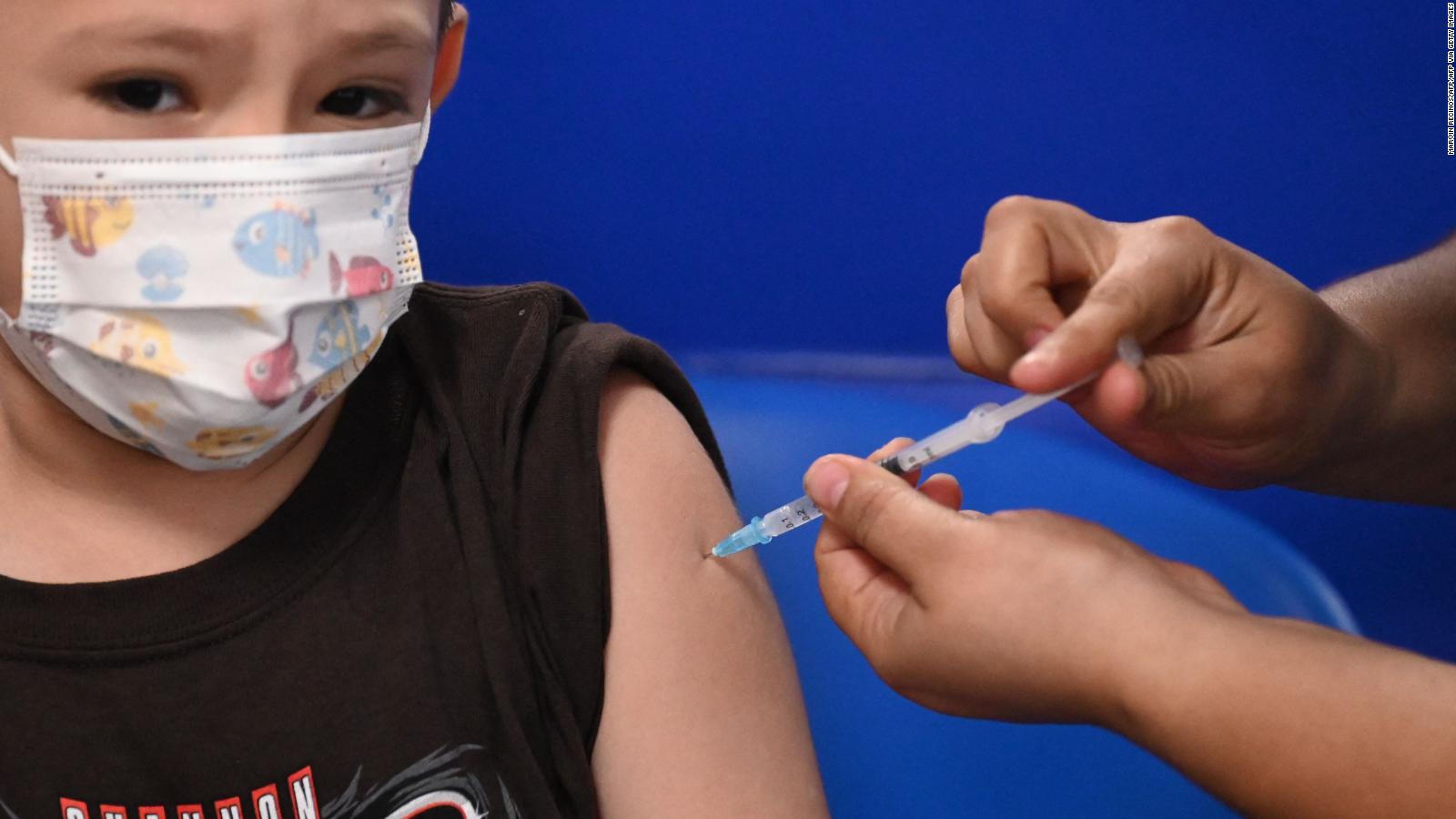
Dr. Andrés Cotton, pediatrician and spokesman for the American Academy of Pediatrics, explained that young children would need to receive a dose of the COVID-19 vaccine three times lower than those over 12 years of age.
Most of the vaccine studies started with adults and a series of safeguards were taken to target younger patients, it adds.
In the United States, it is already anticipated that the Pfizer vaccine could be available for children between 5 and 11 years old by the end of October, after the approval of emergency use that is expected to be delivered by the authorities of the Food and Drug Administration. US Medications (FDA).
Listen to the expert's appreciation.