By Matthew Perrone and Lauran Neegaard -
The Associated Press
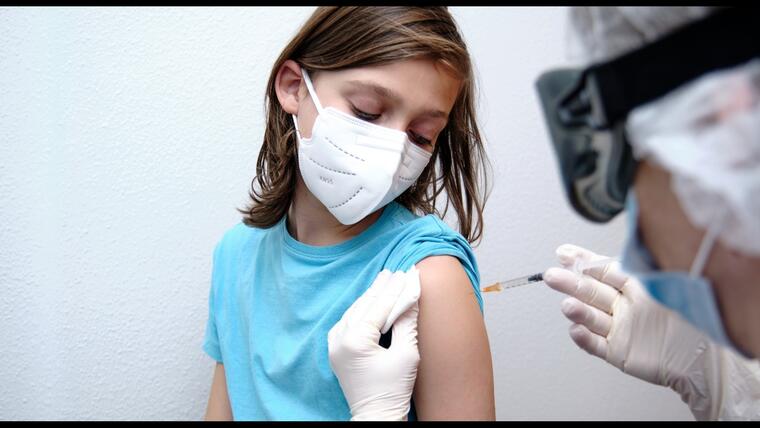
A committee of independent medical advisers decided unanimously on Wednesday to recommend to the Food and Drug Administration (FDA) that it approve the booster dose for the Johnson & Johnson pharmaceuticals coronavirus vaccine.
This second injection should be given at least two months after the first.
The Johnson & Johnson vaccine, unlike Moderna or Pfizer, is administered with a single dose instead of two, but a study presented by the company concluded that protection is triggered if another is administered.
Johnson & Johnson has asked the FDA to give it flexibility on when that second dose should be given, since it might work better if given six months after the first, although studies are still missing to prove that this is the case.
FDA advisers cited this Friday in their decision that growing concern that patients immunized with the Johnson & Johnson vaccine are less protected than those who received the Moderna or Pfizer vaccine, because many of them also received the injection ago. already many months.
In any case, the decision now goes to the FDA, and then the Centers for Disease Control and Prevention (CDC) must decide as well.
[No, Pfizer and Moderna vaccines for children are no more deadly than COVID-19]
They ask states to prepare to immunize children against COVID-19 if Pfizer's vaccine is approved
Oct. 13, 202100: 30
The FDA expert committee yesterday also recommended the Moderna booster dose for the vaccine for the elderly, adults with health problems and workers at risk.
As in the Johnson & Johnson case, the final decision remains for the FDA to make, and then for the CDC to make the final decision.
Federal health authorities did authorize the third dose of the Pfizer-BioNTech vaccine last month for the elderly and adults with health problems, essential workers and individuals exposed to contracting the coronavirus.