(Credit: Scott Heins / Getty Images)
(CNN) -
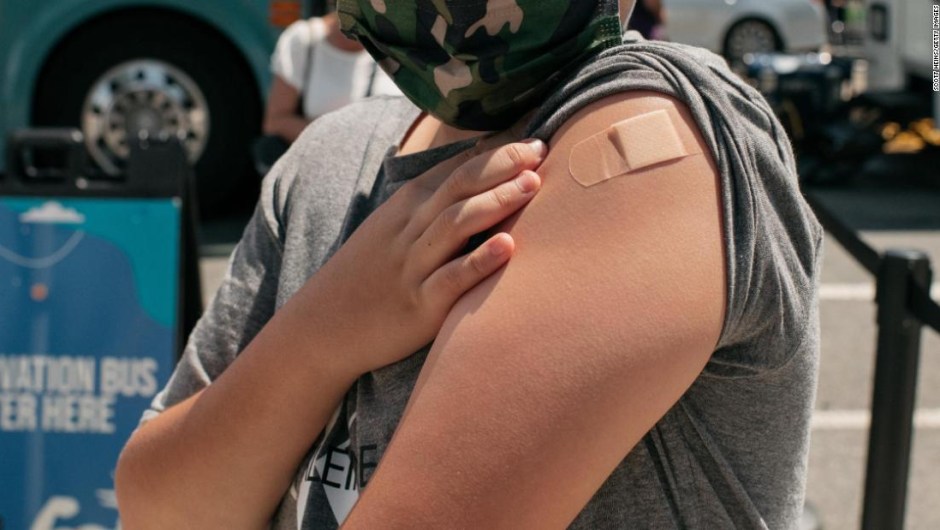
Vaccine advisers from the U.S. Food and Drug Administration (FDA) voted to recommend authorization for emergency use of the one-third-dose vaccine. from Pfizer for children ages 5 to 11.
Members of the FDA's Advisory Committee on Vaccines and Related Biologics agreed that the benefits of vaccinating younger children appeared to outweigh the risks, but some members were concerned about voting to vaccinate a large population of older children. small from studies of a few thousand.
FDA will now consider the committee's vote.
Then, vaccine advisers from the U.S. Centers for Disease Control and Prevention (CDC) will meet next week, Nov. 2-3, to discuss the decision and decide whether to recommend that US children should be vaccinated.
The final word will be on CDC Director Dr. Rochelle Walensky, and vaccination could begin next week if she gives the go-ahead.
Pfizer says its vaccine is 90.7% effective against symptomatic COVID-19 in children ages 5 to 11
With information from Maggie Fox
InstaNews