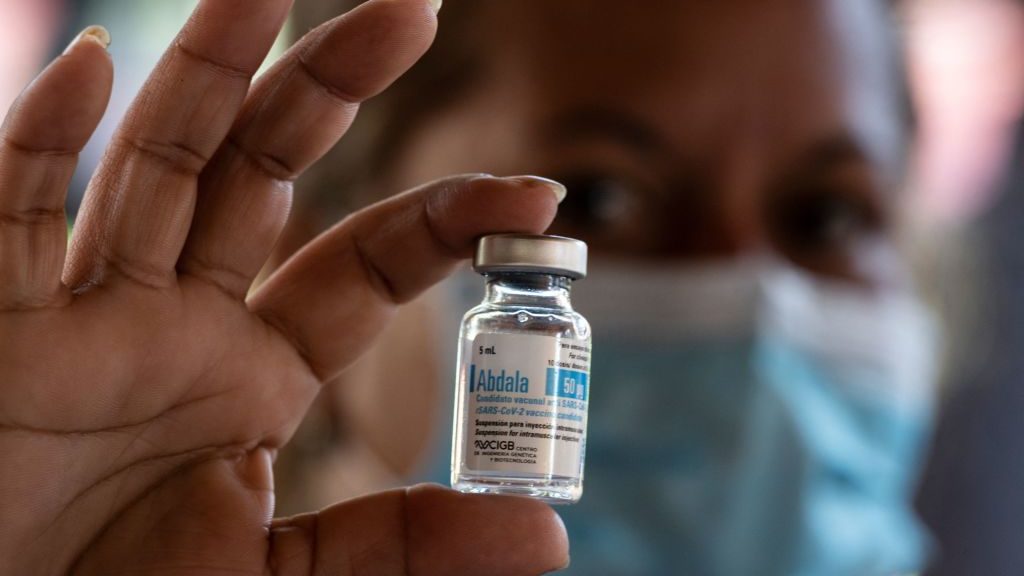
Cuban vaccine is 92.2% effective, according to scientists 0:50
(CNN Spanish) -
The Federal Commission for the Protection against Sanitary Risks of Mexico (Cofrepis) announced this Wednesday the authorization of the emergency use of the Cuban vaccine Abdala.
Through a statement, Cofrepis detailed that specialized personnel carried out the technical-scientific review of the vaccine and determined that it meets the requirements of quality, safety and efficacy.
The Abdala vaccine was developed by the Center for Genetic Engineering and Biotechnology (CIGB) and produced by BioCubaFarma, the state biopharmaceutical group.
A large number of Latin American countries have higher vaccination rates than Europe and the United States
Cofrepis also clarified that the Committee for New Molecules (CMN) met on the use of this biological agent, which received a favorable technical opinion from the experts.
He added that the authorizations issued by the Commission are part of the National Health Regulation Strategy, which allows reviewing and giving access to the greatest number of health supplies, as long as the quality, safety and efficacy of the product are verified.
Mexican doctors receive booster vaccine against covid-19 4:09
For its part, on its Twitter account, BioCubaFarma clarified that with this approval, Mexico can import the Abdala vaccine, for use in the National Vaccination Policy against the Sars-CoV-2 virus, in people over 19 years of age.
advertising
New Year Mexico: these are some of the resolutions of Mexicans for 2022
So far the vaccines that Cofrepis authorizes in Mexico are: Pfizer-BioNTech, AstraZeneca, CanSino Biologics, Sputnik V, Sinovac, Covaxin, Janssen (Johnson & Johnson), Moderna, Sinopharm and, from now on, Abdala.
In addition to Cuba and Mexico, the Abdala vaccine has been licensed in Vietnam, Iran, Venezuela, and Nicaragua.
Coronavirus vaccine