By Matthew Perrone —
The Associated Press
The Food and Drug Administration (FDA) released draft guidelines on Friday that remove
the current three-month abstinence requirement
for donations from men who have sex with other men (gays and bisexuals).
Instead, they would be assessed with a questionnaire that
assesses their individual risks
of contracting HIV based on sexual behavior, recent partners, and other factors.
This policy would also apply to women who have sex with gay or bisexual men.
If this guideline is finally approved, it would not only alleviate discrimination against the LGBTQ community, but it could also bolster blood donations in the United States by increasing donors.
A lifetime ban
The LGBTQ community has long opposed these blanket restrictions on who can donate blood, claiming they are discriminatory.
The American Medical Association and other similar organizations have asserted that such exclusions are unnecessary due to technological advances in testing blood for infectious diseases.
[Over 100 Bills Target LGBTQ Rights]
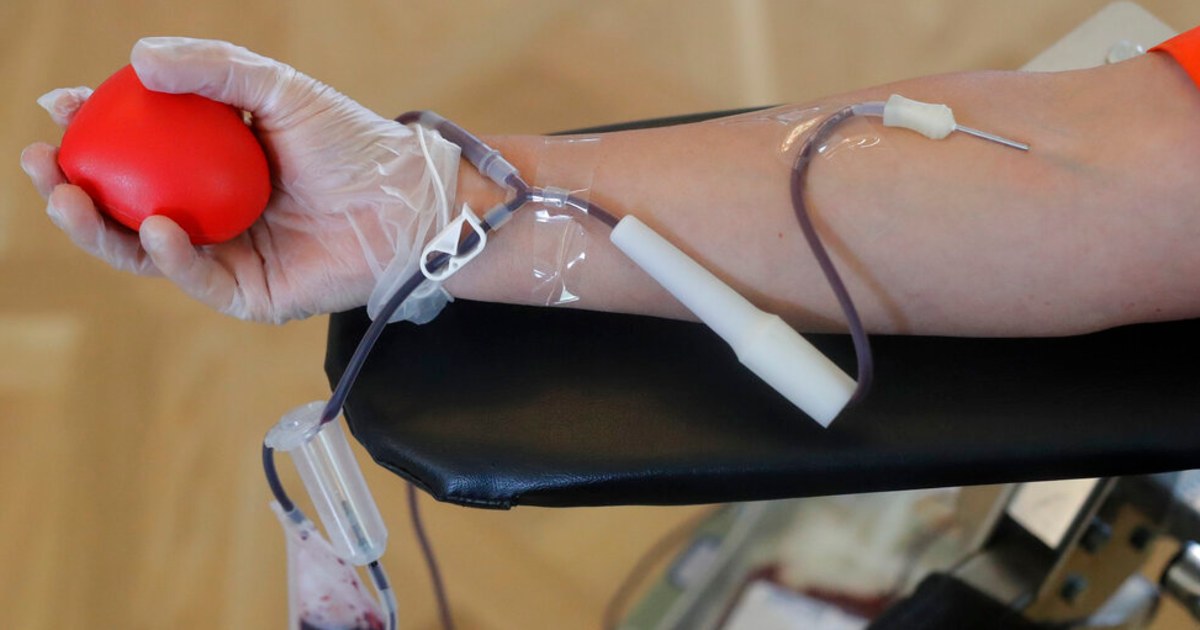
A patient donates blood in California.
Chris Carlson/AP
"Current and previous blood donation policies
made unfounded assumptions
about gay and bisexual men and really entangled people's identity with their likelihood of having HIV," says Sarah Warbelow of the Human Rights Campaign, an LGBTQ rights group. .
The United States and other countries began blocking blood donations from gay and bisexual men
during the AIDS epidemic of
the early 1980s to prevent the spread of HIV through the blood supply, despite the fact that HIV is spread by the same among heterosexual people.
In 2015, the FDA removed the outright ban on these men from donating blood and replaced it with a one-year abstinence requirement.
In 2020, it shortened the period to three months, after donations plummeted during the COVID-19 pandemic.
Regulators say there has been no negative impact on the healthy blood supply as a result of these changes.
The new requirements to donate
All donors answer questions about their sexual history, drug use, and any recent tattoos or piercings, among other factors that may contribute to the spread of blood-borne infections.
The donated blood is then tested for HIV, hepatitis C, syphilis, and other infectious diseases.
They announce that an experimental vaccine against HIV is showing good results against the virus
Dec 2, 202200:30
Under the new questionnaire, men who have sex with men will be asked about new or multiple partners in the last three months.
Those who answer yes to any of the questions and also report anal sex will not be able to donate until a later date.
The policy would also apply to women who have sex with gay or bisexual men.
[Yes, people vaccinated against COVID-19 can donate blood without problems]
This policy mirrors those implemented by Canada and the United Kingdom.
The FDA also based its latest proposal on a recent study of 1,600 gay and bisexual men.
FDA-funded research compared the effectiveness of a detailed, personalized questionnaire on sexual behavior with current time-based abstinence rules.
When will it be effective?
It will take several months for blood banks to make the changes, according to Cliff Numark, an executive at the Vitalant center, who participated in the study.
Changes will require new questionnaires, staff training, and software upgrades.
The Red Cross said it supports the FDA's changes, but added that it's too early to tell if it will result in more blood donations.
The Red Cross faces a crisis in its blood banks.
This is one way to help
Jan 13, 202202:55
Lukas Pietrzak, a Washington DC resident, said he volunteered for the FDA study.
He credits emergency blood transfusions with saving his father's life after a bicycle accident in 1991.
Pietrzak donated blood in high school, but became ineligible after becoming sexually active as a gay man.
“Until I told my friends, I had to avoid explaining why I didn't go to blood drives with them,” says Pietrzak, 26, who now works for the federal government.
“Now we can be part of it,” Pietrzak said.





/cloudfront-eu-central-1.images.arcpublishing.com/prisa/PZFRUMDJ6JBZXN7VSPI6FPM6DU.jpg)