The Food and Drug Administration (FDA) on Wednesday warned consumers to stop using
Delsam Pharma's Artificial Eye Ointment
over fears of possible bacterial contamination.
The federal agency's call comes just weeks after warning that
EzriCare Artificial Tears
from the same manufacturer, already withdrawn from the market, were linked to one death and at least five cases of blindness.
The FDA said
India-based drugmaker Global Pharma Healthcare Private Limited has agreed to recall
Delsam Pharma's Artificial Eye Ointment
,
which was sold by prescription, from the market.
Noticias Telemundo verified this Wednesday that this last product was not available for sale on the company's official website, or on other e-commerce stores such as Amazon.
Parents are alerted for the use of a baby stroller that caused the death of a child
Feb 10, 202300:28
After the report in early February of a death and several cases of blindness after the use of artificial tears
EzriCare Artificial Tears , the drugmaker also agreed to withdraw
Delsam Pharma Artificial Tears
drops from the market
, due to safety concerns.
The FDA this week issued warnings to doctors and patients to avoid using the three aforementioned products, citing potential bacterial contamination.
The Centers for Disease Control and Prevention (CDC, for its acronym in English) will report in early February an outbreak of a rare strain of the bacterium Pseudomonas aeruginosa that has shown resistance to antibiotics, associated with the use of the
products
for eyes then withdrawn.
[One person has died and three have been partially blind after using these eye drops, CDC warns]
As of February 21, the CDC reported about 58 patients with infections related to the use of these artificial tears.
At least five people lost their vision and another died "due to a bloodstream infection," the health entity reported.
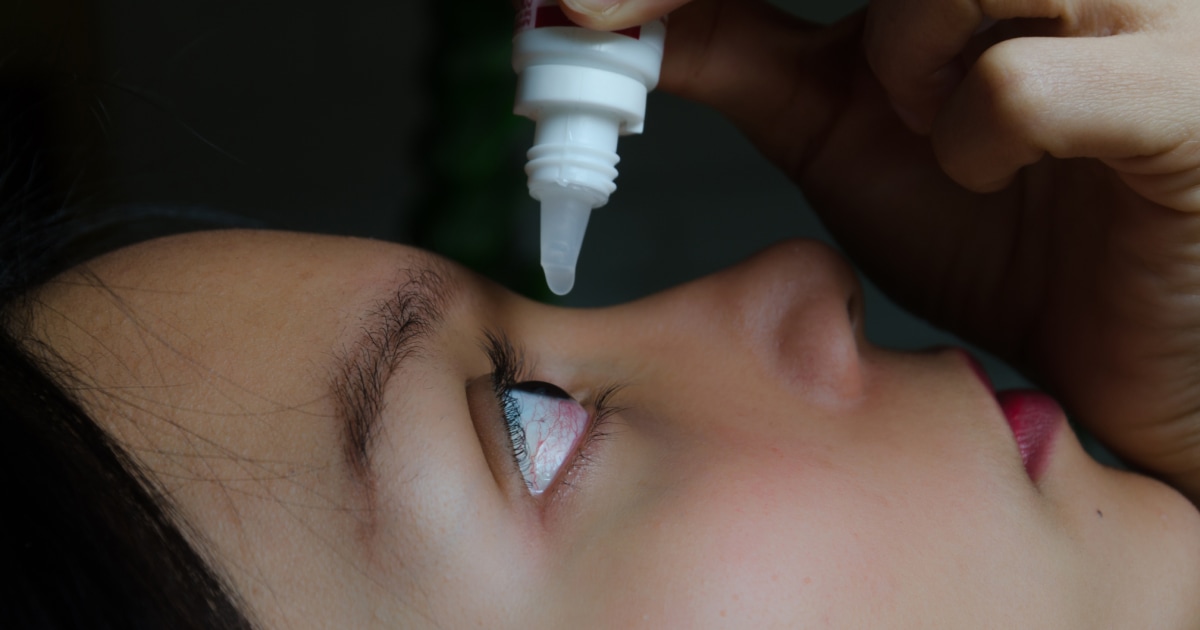
Artificial tears are drops to lubricate dry eyes
and help maintain moisture on the outer surface of the eyes, according to the Mayo Clinic academic center.
“They can be used to treat dry eyes caused by aging, certain medications, a medical condition, eye surgery, or environmental factors, such as smoke or wind,” the entity explains.
This is the brand of baby formula that is withdrawn from the market due to possible contamination
Feb 22, 202300:22
According to the CDC, people who develop symptoms of an eye infection should contact their doctor and seek care immediately.
Some of the warning signs are discharge, pain, discomfort and irritation in the eyes.
Also sensitivity and blurred vision.
The FDA placed an import alert on the pharmaceutical company
Global Pharma Healthcare Private Limited,
which will prevent its products from entering the United States.
The agency stated that the company violated good manufacturing practices due to "lack of proper microbial testing, formulation issues, and lack of appropriate controls regarding tamper-evident packaging."
[Tesla recalls over 360,000 vehicles over safety concerns with "full autonomous driving" feature]
Although patients tested in the outbreak for this bacterium used at least 10 different brands of eye drops,
EzriCare Artificial Tears
was the most widely used among them and the most commonly used among all affected individuals.
According to its website, the pharmaceutical company is present on different continents and its products can be purchased in Latin America.
Delsam Pharma's Artificial Eye Ointment
, the last product to be withdrawn, was recommended for inflammation, infection, burning or dry eyes.
Pseudomonas
bacteria
are found in soil and water.
Its variant
Pseudomonas aeruginosa
causes infections in the blood, lungs and other parts of the body, according to the CDC, which warned that this bacterium is constantly finding new ways to become immune to antibiotics designed to fight it.