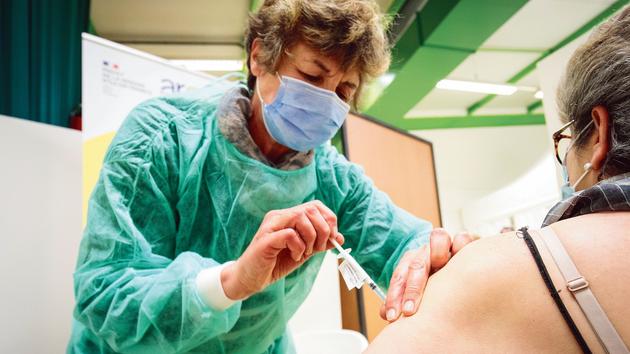
Less than a month after the launch of the French vaccine campaign against Covid, the National Medicines Safety Agency (ANSM) has just published its first pharmacovigilance report.
Put in place for all new treatments arriving on the market, this active monitoring of the possible adverse effects of a drug has been reinforced for vaccines against Sars-CoV-2, due to their faster development than ordinary.
The conclusions of this first stage point are rather reassuring.
"It is a vaccine for which we finally collected very few adverse effects in view of the number of people to whom it was injected"
, analyzes Dr Joëlle Micallef, head of the Marseille pharmacovigilance center, one of the two regional reference centers on the subject in France.
Read also:
Will France be able to vaccinate 70% of adults by the end of August?
The data processed in the report relate to some 388,730 French people vaccinated between December 27 and January 15, mainly with the Comirnaty vaccine from Pfizer-BioNTEch,
This article is for subscribers only.
You have 79% left to discover.
Subscribe: 1 € the first month
Can be canceled at any time
I ENJOY IT
Already subscribed?
Log in