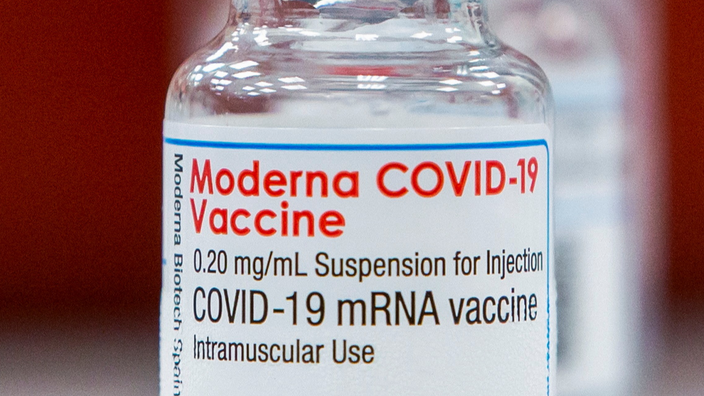
Moderna's vaccine is 90% effective against Covid-19 and 95% against severe forms of the disease, the American biotechnology firm announced in new results published on Tuesday April 13, down slightly from a previous one clinical test.
These data come from a phase 3 clinical trial involving more than 30,000 people in the United States.
Efficacy is down slightly from 94.1% reported in a large clinical trial published in the
New England Journal of Medicine
in December.
To discover
Covid-19: instructions for use of self-tests
Read also: Covid-19: discover the impact of vaccination in your department
Moderna does not specify in its press release whether this drop in efficacy is attributable to the emergence of new variants.
But the biotech firm is working on two modified versions of its variant-specific vaccine, the results of which in mice - not yet peer-reviewed - are encouraging.
“The new preclinical data on our variant-specific candidate vaccines gives us confidence in our ability to anticipate the emergence of new variants,”
applauded Stéphane Bancel, CEO of Moderna.
Read also: Covid-19: can we say that France “produces” vaccines?
As of April 12, Moderna says it has delivered 132 million doses of its anti-Covid vaccine across the planet, including 117 million in the United States.
It remains on track to deliver 100 million more doses to Americans by the end of May, and then 100 million more by the end of June.
The company's clinical trial for adolescents ages 12 to 17 has some 3,000 U.S. participants, and its trial for children ages six months to 11 continues to enroll its 6,750 participants in the United States and China.