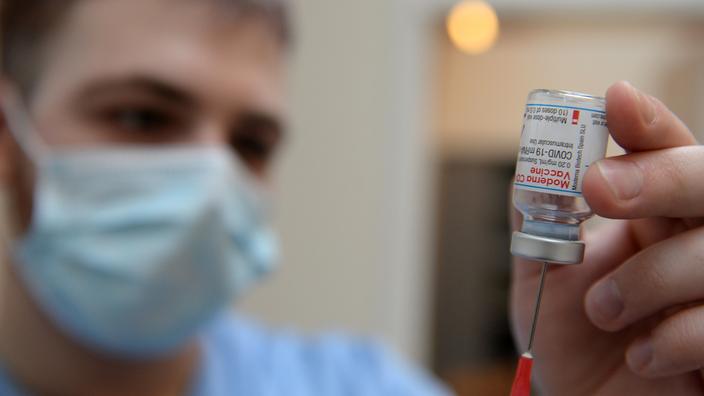
Production of the Moderna vaccine against Covid-19 on French soil will begin "
in March
", Agnès Pannier - Runacher said on RTL.
That of Pfizer will start in April and that of Curevac in May, if it is validated.
These three vaccines will therefore also be "
produced in France
", indicated the Minister of Industry.
France maintains its objective of allowing "
all adults who wish
" to have access to a vaccine by the summer.
Agnès Pannier-Runacher, declared that the executive adapted its plans according to “
good news or bad news
” relating to the epidemic, to the variants as well as to the vagaries of production.
But, she added, "
we are rather confident in our ability to offer French people who wish to be vaccinated by the end of the summer a vaccine
".
To read also: Bertille Bayart: "Vaccines, the fair for false good ideas"
"
Today, as production
started
, we are able to achieve this goal,
" noted the minister, recalling that Pfizer had already caught up with its delay in delivery and that Moderna was on the right track.
Other vaccines could also arrive, "
perhaps earlier than expected
", noted the minister, citing Janssen and Novavax, whose "
results are rather good
".
Yesterday evening, Emmanuel Macron assured for his part that France will be able to offer a vaccine to "
all French adults who wish
", "
by the end of the summer
".
The authorities' objective is now to "
produce more doses faster, and to anticipate the next steps
", for example by producing vaccines "
adapted to the variants
", by the fall.
Other vaccines could be validated by then, such as Russian, which "
can quite find its place
": "
any candidate vaccine which presents a dossier to the AME with all the data necessary for its validation will be examined , as a matter of principle,
”the government member said, adding that the decision was“
scientific, not political
”.