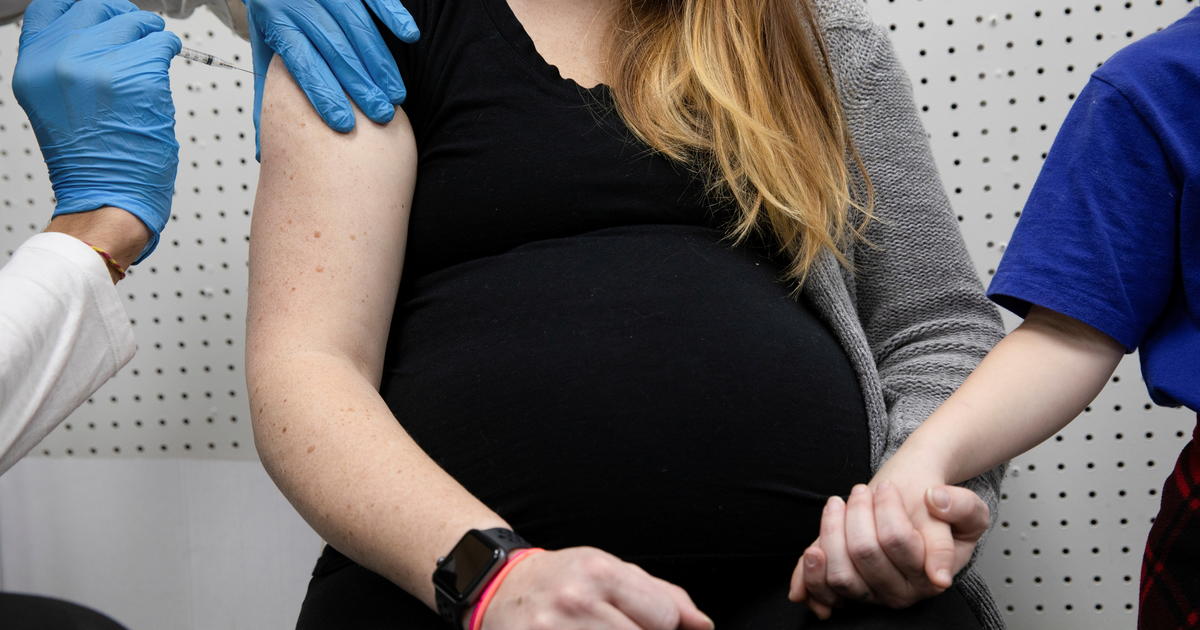
The Centers for Disease Control and Prevention (CDC, in English) rectified on Tuesday a statement made by its director, Dr. Rochelle Walensky, on Friday about the recommendation of the COVID-19 vaccine for pregnant women, and clarified that the agency's orientation has not changed.
A CDC spokesman assured CBS News that its recommendations remain the same as in March, that is, "pregnant people are eligible and can receive the COVID-19 vaccine."
"When faced with the decision of whether or not to receive the COVID-19 vaccine during pregnancy, people should take into account the risk of exposure to COVID-19,
the increased risk of serious infection during pregnancy,
the known benefits of vaccination, and limited but growing evidence on vaccine safety during pregnancy, "added the CDC spokesperson.
Walensky assured during a briefing from the White House that the most recent reports did not show "security concerns" after studying more than 35,000 women in their third trimester of pregnancy, neither for themselves nor for their babies.
"The CDC recommends that pregnant women receive the COVID-19 vaccine," she said, citing a study that found there is no evidence to suggest that the Pfizer and Moderna vaccines pose a risk during pregnancy.
"I encourage people to talk with their doctors and their primary care providers to determine what is best for themselves and their babies," he added.
Specifically, the study cited by Walensky was published last Wednesday in the scientific journal The New England Journal of Medicine, and shows data from more than 35,000 pregnant women who received the Moderna and Pfizer-BioNTech vaccines between December 2020 and February 2021, without experiencing adverse effects.