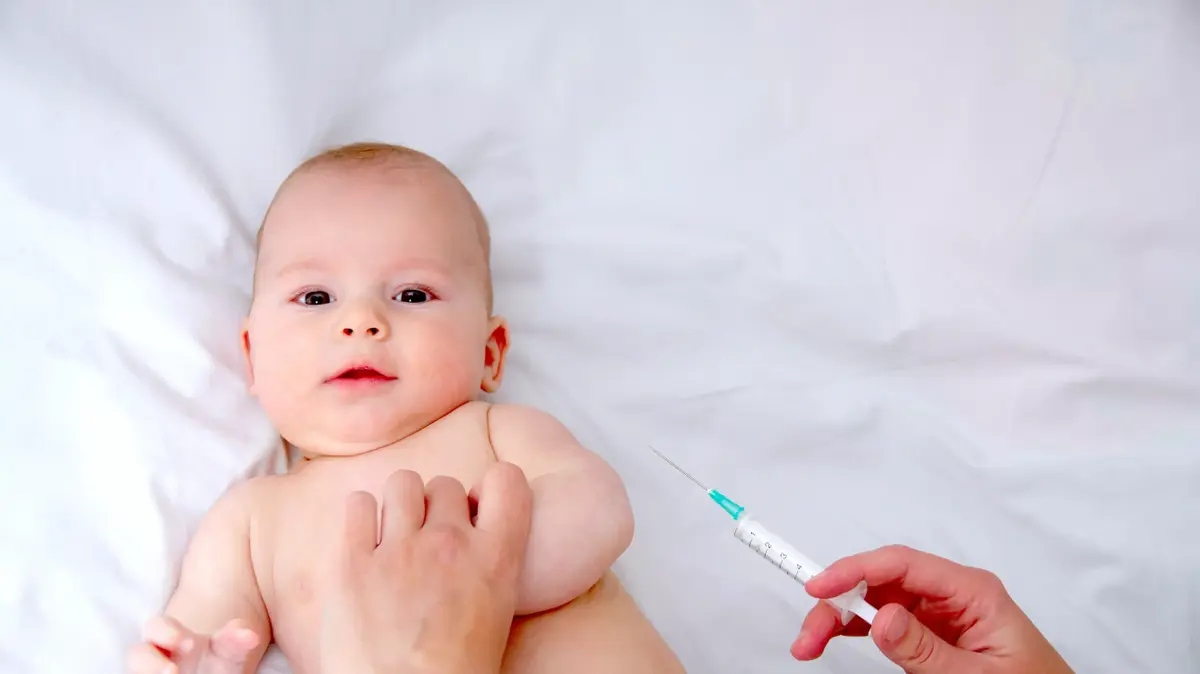
The U.S. Food and Drug Administration (FDA) this evening (Friday) officially approved Pfizer's corona vaccine for use in children ages 5 to 11, after the organization's board of experts voted overwhelmingly in favor of the vaccine.
A tweet on the organization's Twitter page read: "Today we issued an emergency permit for the vaccination against the corona virus from the companies Pfizer and Biontech so that it will also include use among children aged 5 to 11."
The Advisory Committee of Experts of the US Food and Drug Administration decided tonight (Tuesday) to issue an emergency permit for the use of the Pfizer vaccine in 11-5 year olds.
The decision was made by a majority of 17 supporters against one abstention.
Now at the end of the panel's recommendation, the head of the FDA will have to decide whether to accept it or not and then also the US Centers for Disease Control, the CDC.
Pfizer's application sought to grant the vaccine to children on an emergency basis, but in fact it is a request for use for anything and everything until full approval.
The dose of the vaccine for children is actually one-third of the dose of the vaccine for adults.
Similar to the known vaccine, Pfizer's application for children also included the injection of two vaccine doses (adapted for children) at least three weeks apart.
The rationale behind Pfizer's application, despite being highly resistant to the virus, is that vaccination for these ages will allow for the first time in the Western world all preschoolers covered by the Corona vaccine, thus also allowing for morbidity and distribution in schools and other settings.
Also, lowering the infection will help fight the formation of new variants.